Background: Abatacept (Abata, CTLA4Ig), an inhibitor of a key T cell costimulation pathway has recently been approved for GVHD prophylaxis following unrelated donor HCT in combination with CNI/MTX. In 2015, we had developed a novel protocol combining Abata and PTCy for HCT from haploidentical family donors (HFD). We analysed the impact of donor immunogenetics and immune reconstitution on the long-term outcome of 182 patients receiving HFD-HCT on this protocol.
Methods: In patients with nonmalignant diseases (NMD), Abata was administered on days 0, +5, +20, +35 and every 4 weeks thereafter until day +180. PTCy was administered at 50 mg/kg on days +3 and +4 and sirolimus was given from day -7 to 9 months post-HCT (AbaCyS protocol). In those with malignant diseases (MD), PTCy was administered as usual and Abata was administered on day 0 followed by PBSC infusion and on days+7, +21 and +35, followed 6 hours later by donor lymphocyte infusions (DLI). Cyclosporin was administered from day +5 to day +60 (AbaDCyC protocol). NK cell related immunogenetic parameters (KIR genotype/ KIR-ligand/ HLA-B leader peptide dimorphism/ KLRC2 genotype encoding NKG2C receptor, [KLRC2 deletion (del) vs wildtype (wt]) and T and NK cell subset reconstitution were analysed with respect to primary (OS, NRM, Relapse) and secondary (acute & chronic GVHD, CMV reactivation and GVHD & Event-free survival (GVHEFS) endpoints.
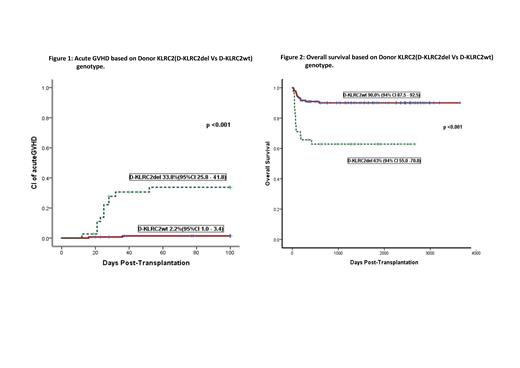
Results: Primary graft failure was seen in 2/46 with NMD and 1/136 with MD, with median time to engraftment being 16 days (12-26). No secondary graft failure was observed. Incidence of aGVHD 2-4 was 8.5%, 4.7% in NMD and 9.8% in MD (p=0.3). Donor KLRC2del (D-KLRC2del) genotype was the only risk factor (33.8%{12/38} vs 2.2%{3/144} in dKLRC2wt, p<0.001, Figure-1) for aGVHD. This translated to a significantly improved NRM (overall 9.5%) in dKLRC2wt group (5.2% vs 28.9% in dKLRC2del, p<0.001). Incidence of cGVHD (mostly mild-moderate) was 15.3% and was not impacted by NMD vs MD or KLRC2 genotype. Relapse incidence (RI) was 21.4%, with no impact of pretransplant disease status (DRI low vs high) or any immunogenetic factors apart from KIR haplotype (lower RI in donor KIR B haplotype, p=0.01). OS was 84.3% at a median follow-up of 4 years, with a significant impact of dKLRC2 genotype (90% in wt vs 63% in del, p<0.001, Figure-2). The overall GVHEFS was 70% and this was also impacted by dKLRC2 genotype (38.2% vs 77.8%in dKLRC2wt, p<0.001). None of the other immunogenetic parameters had any impact on the outcome.
T cell subset reconstitution was robust in both groups, predominantly of memory phenotype, which tended to be more rapid with AbaDCyC protocol in MD. However, recovery of Tregs was more rapid with AbaCyS protocol in NMD and the kinetics of Treg recovery positively correlated with reduced cGVHD in this group. Even though NK cell maturation was brisk in both groups, this was prompter at day+30 in the AbaDCyC protocol group. More importantly, kinetics of NKG2C+adaptive NK cell (ANK) recovery was the only determinant of RI (p<0.01).
Conclusion: The combination of Abata and PTCy was extremely effective in reducing the incidence of aGVHD, which was strongly influenced by dKLRCdel genotype, which in turn adversely impacted NRM, OS and GVHEFS. AbaCyS protocol was associated with better recovery of Tregs vis-à-vis reduced cGVHD. Abata-primed DLI as in AbaDCyC was not associated with increase in GVHD and seemed to moderate the RI in patients with advanced malignancy, which was influenced by ANK recovery. Thus, immunogenetics and immune reconstitution related to ANK subset of NK cells strongly influenced the outcome with Aba-PTCy based GVHD prophylaxis. DLI could be safely administered with Abata priming without increase in GVHD and yet result in attenuation of RI, suggesting the potential of this approach to dissociate GVHD from a NK-mediated GVL effect.
OffLabel Disclosure:
No relevant conflicts of interest to declare.
Abatacept has been used in this study as GVHD prophylaxis and potentiator of NK cell cytotoxicity in Haploidentical HCT.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal